Chromate Information

Chromate:
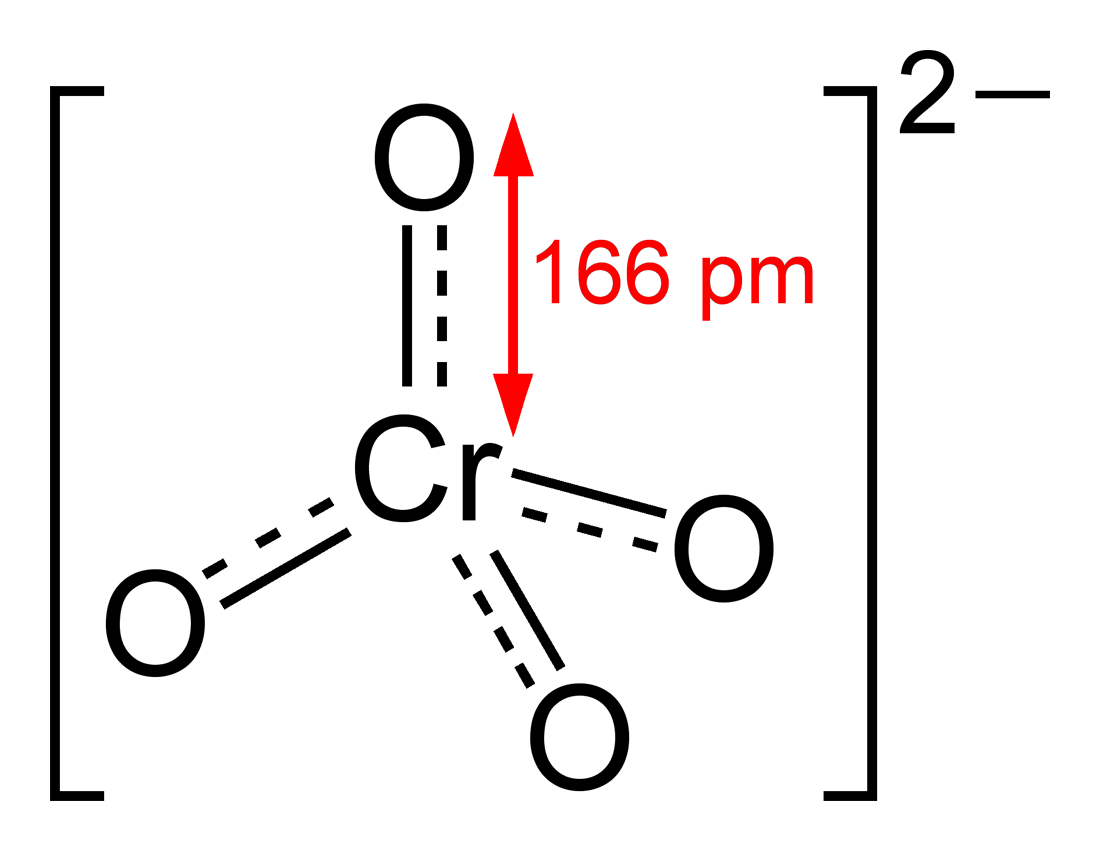
|
Other Names |
Chromate ion, Chromate (VI) |
|
Chemical Formula |
CrO4-2 |
|
CAS Number |
13907 – 45 – 4 |
|
Industry Uses |
Anti-corrosives; Plating; Pigments |
|
Health Risks |
Irritation; Carcinogen |
What is Chromate:
Chromate is an oxoanion of chromium in the 6+ oxidation state. It is a strong oxidizing agent. In aqueous solution, chromate and dichromate are interconvertible. Chromates are used in chrome plating to protect metals from corrosion and improve paint adhesion. Chromate salts are used as pigments, and chrome yellow was a common paint for yellow school buses. Chromate containing minerals are rare. Crocoite is the most commonly found chromate mineral and occurs as long red crystals. Chromates are also used to manufacture leather products, paints, cement, and mortar. They are used extensively as corrosion inhibitors in open and closed cooling water systems.
Chromate Exposure and Health Risks:
All hexavalent chromium compounds (including chromate) are toxic due to their oxidizing power. Chromates are carcinogenic, especially when airborne and cause allergic reactions. Contact with products containing chromates can lead to allergic skin reactions, dermatitis, and ulcers. The use of chromate in manufactured goods has been restricted by the EU (and by market commonality the rest of the world).
Regulations:
The table below summarizes the most-recent standards for Chromium concentration .
|
Limit/Level |
Type |
Organization |
|
0.1 mg/L |
Primary Standards – Max Contaminant Level |
EPA |
Sources: EPA
Measuring Chromate:
Dissolved chromate concentration in water can be measured in units of parts per million (ppm). We carry kits from CHEMetrics that determine chromate levels in water from 0-10 ppm. All of our chromate products can be viewed HERE.
What type of component are you looking for?
| Fixed Mount | Handheld | Dissolved Kits: | Replacement Sensors: | Calibration Gas: | Rentals: |
 |
 |
 |
 |
 |
 |
All sensors require a yearly calibration to ensure your gas measurements are accurate and performing within manufacturer standards. This page is desiccated to the individual manufacturers we represent and their specific calibration procedures.
**Calibration Service Request Form **
|
Calibration costs do vary, see below to get an estimate: Calibration Fee: $150 Analyzer Calibration Fee: $300 PM Calibration Sensor Fee: $330 Genie Calibration Fee: $265 ATI Calibration Fee: $205 ** note that prices are subject to change per labor and parts required. |
Contact us for help choosing the right product for your application