Nitric Oxide Information
Nitric Oxide:
|
Other Names |
Nitrogen oxide; Nitrogen Monoxide; |
|
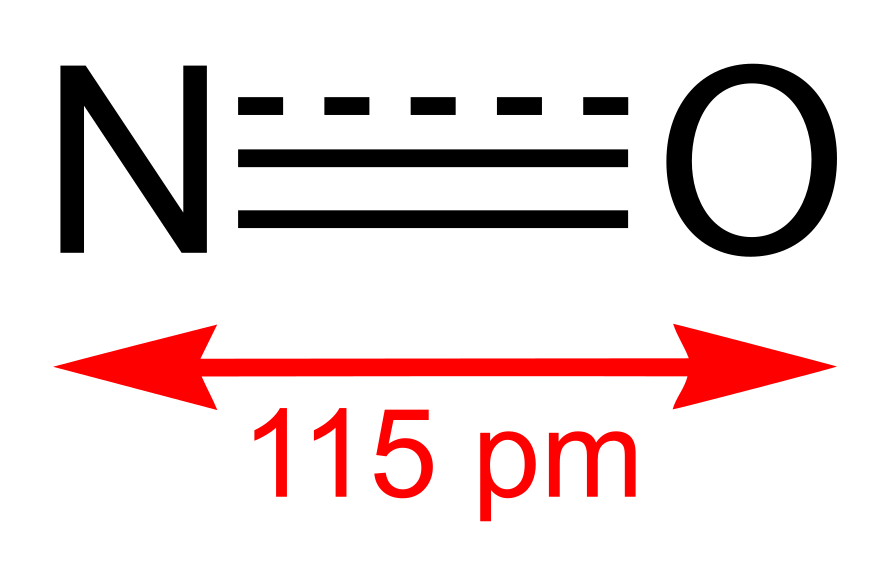
Chemical Formula |
NO |
|
CAS Number |
10102 – 43 – 9 |
|
Industry Uses |
Chemical Laboratories |
|
Health Risks |
Irritation; Organ Damage; Unconsciousness; Death |
|
Vapor Pressure |
34.2 atm |
|
Water Solubility |
Slightly-Soluble |
|
Flammability |
Non-Flammable |
|
Odor |
Sharp, Sweet |
What is Nitric Oxide:
Nitric Oxide is a colorless gas, with a sharp, sweet odor. Nitric Oxide is an unstable, free-radical gas that reacts rapidly with oxygen to form nitrogen oxides. Nitric Oxide is produced in many mammalian tissues and is a mediator of cell-to-cell communication. It functions in many biological processes, most notably vasodilation, inflammation, and neurotransmission. It is noncombustible but as an oxidizer, it accelerates the burning of combustible materials. Heating nitric oxide under pressure will cause it to explode and rocket. Nitric Oxide is very toxic by inhalation and skin absorption.
Nitric Oxide Exposure and Health Risks:
Nitric Oxide is very toxic by inhalation and can be fatal even after a very short exposure to small quantities. It is an irritant to the eyes and respiratory tract. Irritation can cause burning and coughing. Labored breathing, organ damage, and unconsciousness may occur hours after exposure. Low concentrations can be dangerous even for a short time. High concentrations can be fatal after a very short time.
Regulations:
The table below summarizes the most-recent exposure limits.
|
Limit/Level |
Type |
Organization |
|
25 ppm |
TWA (8 hrs) |
OSHA |
|
25 ppm |
TWA (8 hrs) |
NIOSH |
Measuring Nitric Oxide:
Nitric Oxide concentration in the air can be measured in units of parts per million (ppm). Many of our products have digital communication capabilities for easy monitoring and control. We carry portable and fixed Nitric Oxide monitors and kits. All of our Nitric Oxide products can be viewed HERE.
What type of component are you looking for?
| Fixed Mount | Handheld | Replacement Sensors: | Calibration Gas: |
 |
 |
 |
 |
All sensors require a yearly calibration to ensure your gas measurements are accurate and performing within manufacturer standards. This page is desiccated to the individual manufacturers we represent and their specific calibration procedures.
**Calibration Service Request Form **
|
Calibration costs do vary, see below to get an estimate: Calibration Fee: $150 Analyzer Calibration Fee: $300 PM Calibration Sensor Fee: $330 Genie Calibration Fee: $265 ATI Calibration Fee: $205 ** note that prices are subject to change per labor and parts required. |
Contact us for help choosing the right monitor for your application