Thiosulfate Information

Thiosulfate:
|
Other Names |
Thiosulphate; Thiosulfate Ion |
|
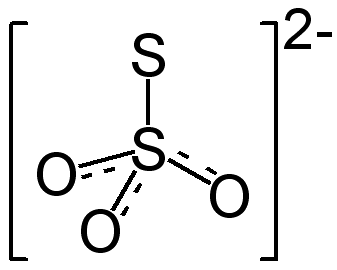
Chemical Formula |
S2O32- |
|
CAS Number |
14383 – 50 – 7 |
|
Industry Uses |
Bleaching; Chemical Laboratories |
|
Health Risks |
Irritation |
What is Thiosulfate:
Thiosulfate is an excellent reducing agent. It is used primarily as a chlorine-removing agent, or antichlor, in various chemical processes. In industry it is used for the bleaching of pulp, paper, and textiles. Thiosulfate Ions are commonly bonded with Sodium to form hyposulfite or sodium thiosulfate. Hyposulfite is used to fix black and white negatives and prints in photography. Ammonium thiosulfate is a fixing salt. In acidic conditions, thiosulfate causes rapid corrosion of metals. It causes pitting corrosion to steel and stainless steel. Thiosulfate occurs naturally only in a very rare mineral, sidpietersite.
Thiosulfate Exposure and Health Risks:
Thiosulfate may cause skin and eye irritation if contacted directly. Prolonged or repeated contact can cause dermatitis. Inhalation of thiosulfate can cause respiratory distress and irritation of the entire respiratory tract. Ingestion of thiosulfate may be harmful.
Regulations:
There are no explicit restrictions or regulations from the EPA regarding Thiosulfate concentration in water.
Measuring Thiosulfate:
Dissolved Thiosulfate concentration in water can be measured in units of parts per million (ppm). We carry instrumental kits from CHEMetrics that determine thiosulfate levels in water. All of our thiosulfate products can be viewed HERE.
What type of component are you looking for?
| Fixed Mount | Handheld | Dissolved Kits: | Replacement Sensors: | Calibration Gas: | Rentals: |
 |
 |
 |
 |
 |
 |
All sensors require a yearly calibration to ensure your gas measurements are accurate and performing within manufacturer standards. This page is desiccated to the individual manufacturers we represent and their specific calibration procedures.
**Calibration Service Request Form **
|
Calibration costs do vary, see below to get an estimate: Calibration Fee: $150 Analyzer Calibration Fee: $300 PM Calibration Sensor Fee: $330 Genie Calibration Fee: $265 ATI Calibration Fee: $205 ** note that prices are subject to change per labor and parts required. |
Contact us for help choosing the right monitor for your application